Robot-assisted surgery is still perceived as “the future is here,” a hallmark of high-tech medicine, and is on the verge of becoming routine practice. In eye microsurgery, where microscopic precision of movements is critical, robotic surgical assistants are often indispensable. But now Chinese medical researchers have developed and begun testing a robot capable of performing intraocular surgeries autonomously. Soon, we may face a situation where the human surgeon doesn’t control the robot-surgeon at all, but simply observes. Who will bear responsibility for the surgical outcome? Isn’t the risk too high?
Given the stakes, it is hard to argue that the potential benefits do not outweigh the ethical concerns. Diseases of the retina and its blood vessels, such as diabetic retinopathy and retinal vein occlusion, frequently lead to severe vision loss or complete blindness, profoundly impairing quality of life. Age-related macular degeneration (AMD), in particular, is the leading cause of blindness in developed countries. According to current projections, the number of AMD cases worldwide may reach 288 million by 2040, with a substantial proportion of patients at risk of vision loss.
There are no simple treatments for these conditions. Many patients require regular intravitreal injections as part of anti–vascular endothelial growth factor (anti-VEGF) therapy. Others undergo subretinal gene therapy injections for inherited retinal dystrophies or investigational treatments for AMD, while retinal vein occlusion may require delicate cannulation of affected vessels.
Gene therapy is already available for some patients — for example, Luxturna — but delivering it is technically demanding. The drug must be injected into the narrow space between the retinal pigment epithelium and the photoreceptors in the macular region, where the risk of retinal tear or detachment is significant.
Operating in this confined space is extremely challenging for a human surgeon. The retinal pigment epithelium is approximately 25 micrometers thick, and retinal vessels can measure less than 150 micrometers in diameter. To perform a subretinal injection, the needle must reach a layer only tens of micrometers thick — an accuracy that is difficult to achieve consistently without robotic assistance. Physiological hand tremor can cause instrument movements ranging from 14 to 142 micrometers at rest and up to 340 micrometers during active manipulation.
The primary aim of robotic assistants in intraocular surgery has been to suppress physiological tremor, scale the surgeon’s movements, and stabilize delicate instruments. Such systems include the KU Leuven co-manipulator, PRECEYES, and IRISS. In pilot studies, these platforms demonstrated improved trajectory control and reduced tissue damage, particularly among less experienced surgeons.
Yet robotic assistants have inherent limitations. They still require the surgeon to monitor the surgical field, make decisions, and guide the instrument in real time. Maintaining a needle within a vessel measuring just 100–150 micrometers in diameter is technically demanding. The surgeon must simultaneously compensate for respiratory motion, subtle eye micro-movements. Even perfect tremor filtering cannot fully eliminate cognitive load or delays in human reaction time. Moreover, robotic assistants lack global navigation: they only see what is within the microscope’s field of view and do not build a dynamic three-dimensional map of the entire eye. In this sense, they function primarily as extensions of the surgeon’s hand.
A team of Chinese researchers led by Gui-Bin Bian has taken a further step toward autonomy. Their system, ARISE, described in Science Robotics, is designed to perform complex retinal injections — including subretinal and intravascular procedures — without direct human control, achieving a level of precision beyond that of human hands or conventional telemanipulators.
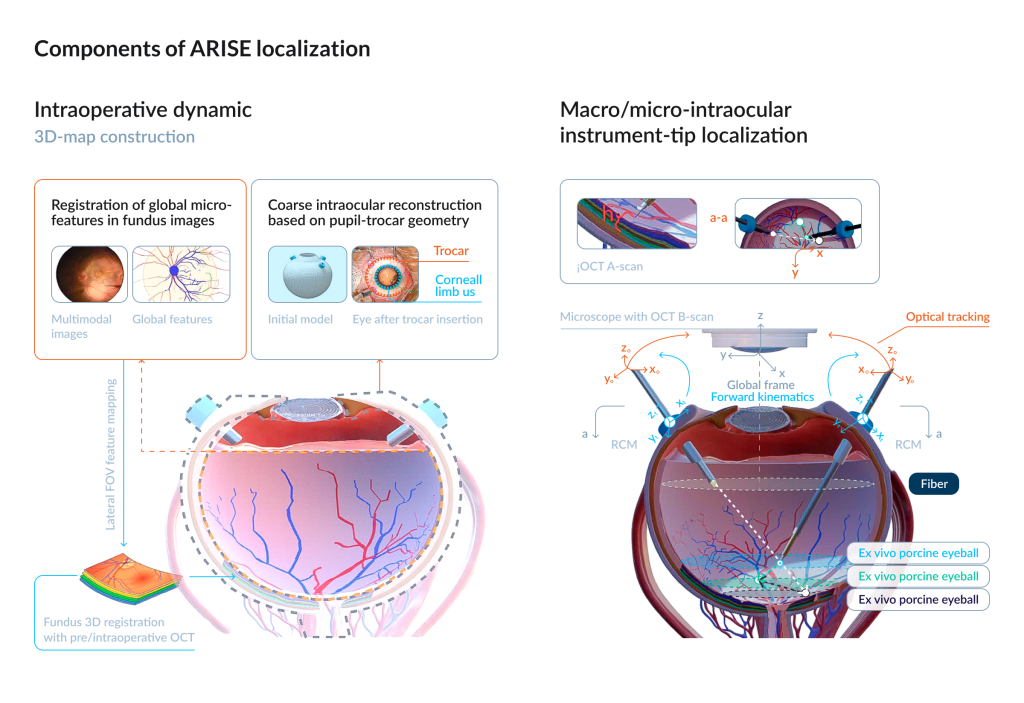
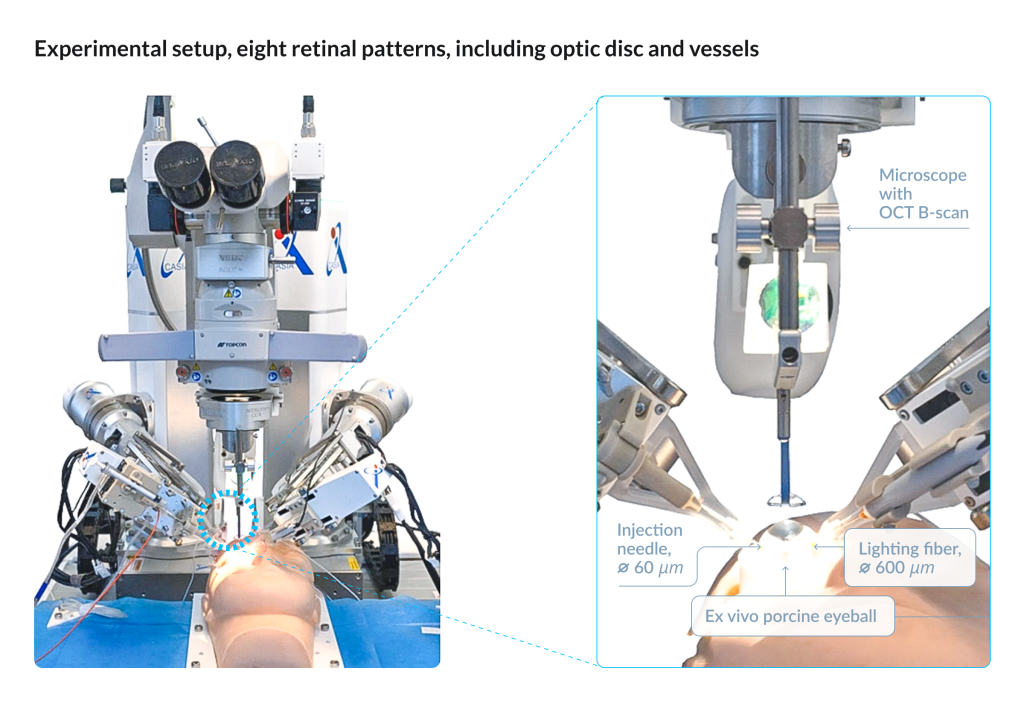
ARISE is a dual-manipulator microsurgical robot. The right manipulator holds an injection needle equipped with force and distance sensing, whereas the left manipulator holds a medical lighting fiber for intraocular illumination. It offers 12 degrees of freedom, micron-level positioning accuracy, and an integrated navigation and safety control system. The platform constructs a dynamic three-dimensional model of the eye by integrating data from the surgical microscope, preoperative and intraoperative optical coherence tomography (OCT), optical tracking systems, and onboard position sensors.
Gui-Bin Bian et al. / Science Robotics, (2026)
For precise needle guidance, ARISE relies on two algorithmic layers. A multiview spatial fusion module aligns multimodal images and compensates in real time for tissue deformation and optical shifts. A criterion-weighted sensor fusion layer integrates kinematic data, optical tracking, visual input, intraoperative OCT, and force sensing to determine the needle tip’s position at both macro- and micro-scales.
The procedure begins with the surgeon defining the surgical target — a specific point on the retina or within a vessel. From there, the robot proceeds autonomously: entering through the trocar, identifying the operative field, calibrating its position, advancing the needle, and performing puncture and injection along a planned trajectory. During the procedure, the system continuously monitors forces at the needle tip, visual indicators of tissue penetration, and depth limits. It also generates alerts in response to atypical tissue behavior, allowing the surgeon to assume immediate control if necessary.
The system has not yet entered clinical trials. Its performance has been evaluated in silicone eye phantoms, ex vivo porcine eyes, and in vivo pig and rabbit models. In both ex vivo and in vivo experiments, the robot achieved a 100 percent technical success rate across three injection types: subretinal, central retinal vein, and branch retinal vein.
Compared with manual and robot-assisted approaches, autonomous operation substantially reduced positioning errors. Mean axis errors decreased by 55 to 80 percent, with the most pronounced improvement observed along the depth (z) axis, where the system achieved micrometer-scale precision.
In the Nature review, clinicians and engineers argue that procedures such as subretinal injection and retinal vein cannulation are particularly well suited to robotic systems. These tasks demand sub-millimeter precision and prolonged instrument stability — capabilities that challenge even highly experienced surgeons.
Picture granted by Gui-Bin Bian.
The developers of ARISE describe the system as a means of increasing procedural repeatability, reducing reliance on individual skill, and enabling surgeons to shift from manual execution to oversight and decision-making.
But the move toward autonomy raises several questions. Experimental models cannot fully replicate the variability of human pathology. There is no certainty about the consequences of possible algorithmic error and about system performance in rare anatomical variants. Recent reviews emphasize that such platforms will require pragmatic clinical trials powered to detect differences in clinically meaningful outcomes.
Even if an autonomous robot can hold a needle more steadily than a human surgeon, micrometer-scale precision is not the ultimate measure of success. What ultimately matters is whether the technology improves visual outcomes and quality of life — and whether it does so within acceptable limits of risk, operating time, and cost.
The future of autonomous robotic surgeons will be determined by clinical trials. These trials have to demonstrate not only a reduction in postoperative complications but also a significant improvement in the accessibility of technically demanding procedures.


