Aging had long appeared to be a slow wear and tear of everything at once. As we previously mentioned, it is increasingly described today as a set of specific malfunctions that can be measured and partly corrected in animal experiments. This article looks at what can already be done about aging associated with such hallmarks as protein quality control, mitochondrial function (the cell’s power plants), and the accumulation of senescent cells – those that have stopped dividing and functioning for the benefit of the organism yet continue to damage the surrounding environment. These processes reinforce one another.
Loss of protein quality control: when the assembly line produces defects
Proteins are the working tools of the cell. They need to adopt a precise three-dimensional structure to function properly. When folding goes wrong, misfolded proteins can accumulate and disrupt normal cellular processes. Cells maintain order in two ways: helping proteins complete their proper folding and rapidly grinding up what can no longer be saved. With age, this infrastructure weakens, and damaged proteins begin to accumulate as aggregates – a typical finding in many age-related diseases of the nervous system.
The proteasome is one of the key complexes responsible for breaking down damaged proteins. If waste processing is accelerated, tissue remains functional for longer. In experiments on the nematode C. elegans, enhancing proteasome activity extended lifespan: the average lifespan increased by more than a quarter – from 14.0 to 17.7 days. Another striking marker was that they had less age-related pigment in the intestine – an indicator of waste accumulation, meaning aging had progressed more slowly.
A similar effect was observed in fruit flies. When the activity of a key proteasome component was increased in adult Drosophila, older flies had fewer protein deposits, and the average lifespan increased by 10–15% in repeated experiments.
This is important because even only targeting the process of protein quality control potentially affects many diseases at once. When waste accumulates, almost everything malfunctions – from neurons to muscles. That is why loss of protein quality control stands among the fundamental hallmarks of aging alongside mitochondria and senescence.

Mitochondrial dysfunction: power plants produce less energy and more smoke
Mitochondria are the energy stations inside cells. They produce energy for movement, thinking, and immune defense. With age, some of these stations begin to function worse: there is less energy, more collateral damage, and tissues tire more quickly and recover less effectively.
In one experiment in mice, researchers created transgenic animals whose mitochondria retained their functionality for a longer period. The average lifespan increased by 17% in one type of mouse and by 21% in another, corresponding to approximately +4.5 months and +5.5 months. The maximum lifespan also increased by 10%. The study also found a slowing of age-related changes in the heart and later development of cataracts in such mice.
A second route to the mitochondrial problem involved removing broken organelles. Researchers used the compound urolithin A, which triggered mitochondrial renewal in nematodes, prevented the accumulation of defective mitochondria, and extended lifespan; the worms also maintained mobility and pharyngeal pumping function for longer – direct indicators of vitality in this model. In experiments on rodents, the authors reported improvements in physical performance when measuring age-related decline in muscle function.
This approach was also tested in humans. In a randomized clinical trial in adults aged 65–90, supplementation with urolithin A improved muscle endurance, measured by how many repeated contractions a person was able to perform before suffering from fatigue. It also positively altered certain biomarkers. However, the key primary endpoints in this protocol did not demonstrate a statistically significant effect.
Cellular senescence: zombie cells
Senescent cells, which are also called zombie cells, stop dividing after stress and damage but remain in the tissue. They secrete a mixture of signals that sustain inflammation and impair the function of neighboring cells, which then become senescent more rapidly themselves. With age, the number of such cells increases, and this is considered an independent hallmark of aging.
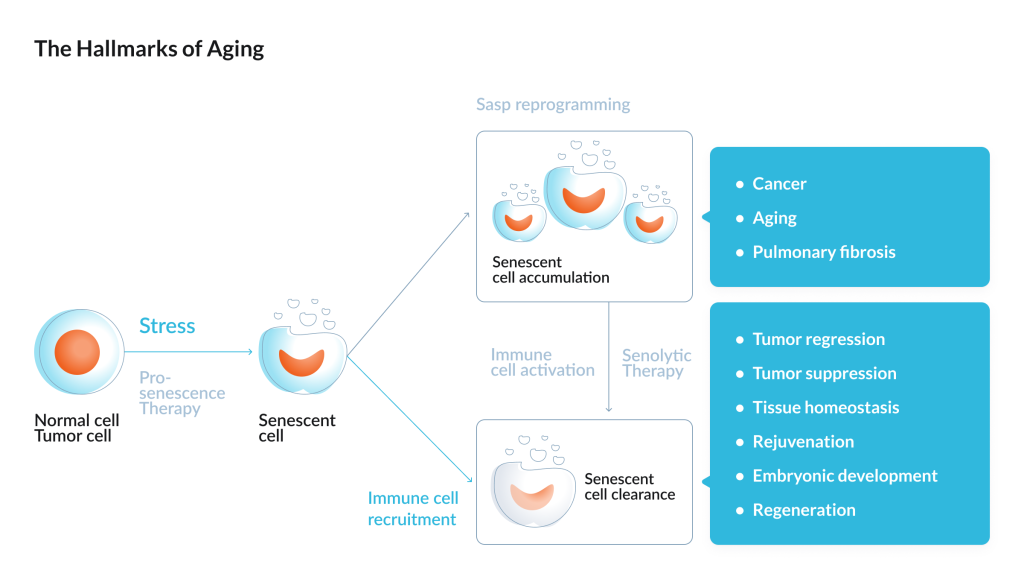
The Mayo Clinic group showed a direct causal effect in mice: if senescent cells are regularly removed, the animals show an extension of average lifespan, later development of tumors, and weaker manifestations of age-related decline in several organs, including the kidneys, heart, and adipose tissue.
An active area of work is senolytics – drugs designed to selectively clear senescent cells. In a Nature Medicine publication, it was shown that transplantation of even a small number of senescent cells into young mice caused persistent deterioration of physical function. Periodic treatment witha combination of dasatinib plus quercetin, which eliminated senescent cells, reduced manifestations of physical frailty and increased survival after treatment by 36% in those with transplanted senescent cells and in naturally aged mice.

This line of research has already reached the clinic. In a pilot study in patients with diabetic kidney disease, a short 3-day course of the combination of dasatinib plus quercetin was accompanied by a reduction in markers of senescent cells, as shown in biopsies of adipose tissue and skin. The intervention also led to a decrease in several circulating inflammatory factors.
Why is this important?
Together, these mechanisms help explain why aging accelerates across many tissues. Improving protein quality control, mitochondrial performance, and the clearance of senescent cells can preserve tissue function longer in experimental models. In an article from 2023, these processes are described as interconnected nodes of aging that can already be targeted experimentally.
In animals, measurable effects are already being achieved – sometimes in lifespan, sometimes in organ function and endurance. The next barrier is safety and reproducibility in humans: the mechanisms that protect us from cancer and damage must not be interfered with crudely. Therefore, the main currency in the biology of aging today is careful engineering and high-quality clinical measurement.


