Most age-related diseases — from cancer to neurodegeneration — connect back to one core problem: over time, cells get worse at storing their DNA safely and using it correctly. In the “Hallmarks of Aging” framework(which we discussed here), researchers highlight three aging processes that sit right on top of our genetic material: DNA damage, telomere shortening, and epigenetic drift, when the settings that control gene activity get noisy.
In January 2026, this field crossed a new milestone: the US drug regulator FDA cleared an IND for Life Biosciences’ ER-100, meaning the agency allowed the company to start a first-in-human clinical trial of a partial epigenetic reprogramming gene therapy in people. The goal is to treat diseases where the optic nerve is damaged and vision declines. ER-100 aims to shift the gene-control settings in retinal cells toward a healthier, more youthful pattern — without rewriting a person’s DNA sequence.
In this article, we explain how these processes unfold, what animal studies have shown so far, and what to realistically expect from early human testing in 2026–2027.
Genomic instability: “typos and damage” in the DNA instruction book
Imagine DNA as a giant instruction manual. Every day, pages get scuffed: sunlight, normal metabolism, inflammation, and plain copying errors can all leave small chemical injuries or even breaks in the DNA strand. Young cells usually repair a lot of this damage, but with age the repair systems become less reliable. When damage piles up, cells may malfunction, shut down, die, or accumulate mutations that raise the chance of becoming cancerous.
A concrete animal example involves NAD⁺, a molecule cells use for energy chemistry and for certain repair proteins. In a Science paper, researchers showed that as mice age, falling NAD⁺ levels can interfere with a key DNA-repair route involving the protein PARP1. When they raised NAD⁺ in older mice, some molecular readouts moved in a more youthful direction: PARP1 activity increased and DNA-damage markers dropped.
DNA repair is hard to turn into a safe anti-aging therapy. These systems sit close to cancer biology: pushing cells too hard to divide or overriding safety brakes can backfire. So the near-term goal would be to identify which repair pathways fail first and in which tissues, then figure out how to support them without raising tumor risk.
Telomere attrition: the “plastic tips” on chromosome shoelaces
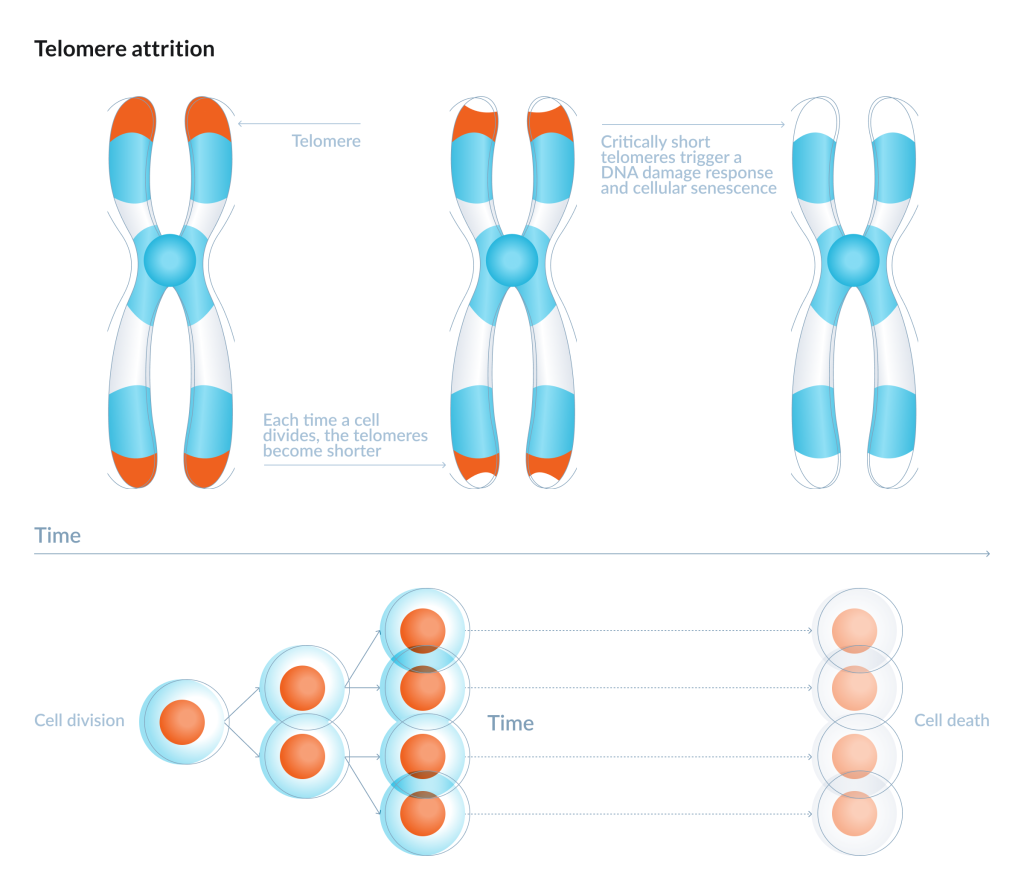
Telomeres are protective caps at the ends of chromosomes — like the plastic tips on shoelaces. Each time a cell divides, telomeres tend to shorten. When they become too short, the cell is more likely to stop dividing or become dysfunctional. For tissues that need renewal, that looks like a repair crew running out of tools.
A direct approach is telomerase, an enzyme that can lengthen telomeres. In an influential mouse study, researchers used an AAV vector to deliver the telomerase gene (TERT) to adult and old mice. They reported that median lifespan increased by 24% in 1-year-old mice and 13% in 2-year-old mice, alongside improvements in several “healthspan” measures such as insulin sensitivity, neuromuscular coordination, and other aging-linked biomarkers. In this study, cancer rates did not increase versus controls.
Even so, safety concerns remain. Telomerase activity is a well-known feature in many cancers, because it helps cells keep dividing. Any human therapy that targets telomeres or telomerase will need strong long-term cancer-risk data.
Epigenetic alterations: when the “reading settings” drift out of tune
If DNA is the text, epigenetics is closer to the reading settings: bookmarks, highlights, and rules that tell the cell which genes to turn on, which to silence, and when. Over time those settings become noisy. Some genes get turned up too much, others go quiet, and cells gradually lose their precise identity and performance.
Full cellular reprogramming into stem-like cells (iPSCs) can wipe away many molecular signs of age, erase cell identity, and raise tumor risk. That’s why partial reprogramming became the more practical goal: shift cells toward a younger “settings profile” while keeping them as the same kind of cell.
One widely discussed tool here is the Yamanaka factors — proteins that act like powerful gene-control switches. In the ER-100 story, the key trio is OSK, short for Oct4, Sox2, and Klf4. These proteins can change how cells run their genetic programs and, under controlled conditions, move epigenetic patterns toward a more youthful state without changing the DNA sequence itself.
In a Nature paper, researchers made retinal cells in mice temporarily produce OSK and reported three key findings: the cells’ gene-control patterns shifted toward a more youthful state, injured nerve fibers regrew better, and vision loss was partly reversed in a mouse glaucoma model and in aged mice.
A related Cell study in mice with accelerated aging used short, repeated “pulses” of OSKM (OSK plus c-Myc) rather than leaving it on continuously. The authors reported improvements in multiple age-linked features and emphasized that the dosing schedule matters for safety, because uncontrolled reprogramming can push cells into unsafe states.
That risk is not theoretical. Reviews and experimental work repeatedly flag the hazards: teratomas if reprogramming goes too far, activation of cancer-related programs, and toxicity when the wrong tissue or dosing scheme is used.
2026: human test begins (ER-100)
On January 28, 2026, Life Biosciences announced that the FDA cleared its IND for ER-100 in optic neuropathies. The clearance allows a Phase 1 safety study to begin.
ER-100 is an eye-directed gene therapy. A modified AAV vector is injected into one eye to deliver instructions that make retinal cells temporarily produce OSK. The system is designed so that OSK expression is activated for a limited time using doxycycline. The trial documentation states that ER-100 leaves a participant’s DNA sequence unchanged.
The study plans up to 18 participants: 12 with open-angle glaucoma and 6 with non-arteritic anterior ischemic optic neuropathy (NAION). NAION is more common after age 50 and is linked to vascular risk factors, and no treatment reliably restores lost vision. The trial starts with dose-finding in glaucoma, then moves to NAION at a selected dose. Doxycycline is given for 8 weeks (56 days), and participants are followed long-term with visits extending up to 5 years, which is typical for gene therapy safety monitoring.

The main endpoints are safety and tolerability, plus immune reactions (including neutralizing antibodies to AAV2). The study also tracks vision and retinal structure using standard eye tests.
In its public statements, the company says the program builds on years of preclinical development and studies in nonhuman primates, with controlled OSK expression and signals consistent with improved visual function. These claims are preliminary; peer-reviewed papers and trial readouts will test them.
What to expect in 2026–2027
In the next one to two years, ER-100 should deliver safety information: local inflammation inside the eye, immune responses to the vector, how well people tolerate the doxycycline activation period, and whether there are any warning signs that cells are being pushed into unsafe programs.
If the safety profile looks acceptable, that alone would be a major milestone: it would establish a real-world playbook for testing partial epigenetic reprogramming in humans. Over time, similar ideas may expand into trials targeting other tissues. Only after the effectiveness and safety of these interventions are proven can we expect access to this type of therapy. Although the path is not a fast one, even the very possibility of influencing how our genetic code is expressed and reversing age-related conditions would have sounded like science fiction 15–20 years ago.


