Cast your mind back 15 years. Someone tells you that stepping out the door, you can take along a blood pressure monitor, a glucose meter, a pulse oximeter, and even an ECG device — and that all of these will run nonstop, mostly sitting on your wrist inside a watch. Would you have bought into that kind of science fiction back then? It turned out that not much time at all was needed for it to become reality. Wearable medical devices that we carry with us every day are evolving at a breakneck pace, surprising us year after year. Today, we’re looking back at the most landmark developments of recent years and checking in on where the industry is headed.
Even the smartwatch on your wrist has clearly moved beyond being just a gadget that entertains you with a stream of data. The information it generates now qualifies as fully valid medical data, encompassing what is essentially continuous monitoring of a wide range of body’s vital indicators.
But the clearest sign that wearables are shedding their identity as fitness toys is the attention they’re drawing from clinics, insurers, employers, foundations, and research groups. The WHO ties advancements in digital health to the strengthening of healthcare systems, while McKinsey’s review from last year points out:
“Gen Z and millennials are more likely to be maximalist optimizers, digitally savvy consumers who experiment with a wide range of health and wellness products and conduct extensive research to find what works for them. This consumer segment represents roughly 25 percent of wellness consumers and more than 40 percent of market spend. They are twice as likely as the overall population to use natural and alternative products, as well as cutting-edge digital technologies and health-tracking devices — if they have confidence that the products will work for them.”
Stars have aligned — there’s strong demand, and companies are ready to keep rolling out new offerings to meet it.
What Are Wearable Medical Devices?
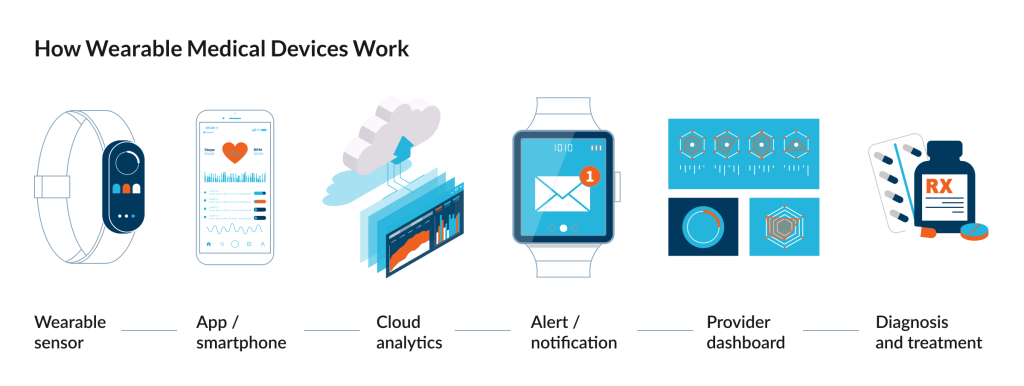
Let’s first sort out the terminology. To put it simply, wearable medical devices are devices worn on the body that collect physiological data outside of a doctor’s office. Sometimes that means a standard smartwatch or fitness band; other times, it’s a specialized wearable sensor, biosensor, or patch that can track heart rhythm, temperature, sleep, respiration, blood pressure, ECG, blood glucose, or other vital signs. The FDA describes these solutions as sensor-based digital health technologies and explicitly points out that they can fall under two categories — general wellness or a regulated medical device with a specific intended use.

But how do you tell a serious, clinically and regulatory-approved device apart from a consumer toy? The difference doesn’t come down to how it looks — it comes down to purpose and evidence. One type of product is built for health and wellness: tracking workouts, sleep, heart rate, and daily habits. The other is designed for diagnostic or monitoring scenarios: cardiac surveillance, diabetes management, epilepsy, post-hospitalization follow-up, or rehabilitation.
In one case, the user looks over their own numbers. In the other, a provider reads the data and makes downstream decisions about diagnosis, care pathways, and treatment. That’s exactly why the wearables market looks so varied right now. The single word “wearables” covers everything from Apple Watch and Garmin to CGM systems, exoskeletons, and clinical patches for continuous monitoring. It’s easy to get lost in the mix if you don’t have a clear enough grasp of the underlying logic.
Why Wearable Medical Devices Matter in Health Tech?
The surge in demand is no accident. According to WHO data, noncommunicable diseases — cardiovascular conditions, cancer, diabetes, and chronic lung disease — account for roughly 74% of all deaths worldwide. It stands to reason that early detection of dangerous conditions and continuous monitoring of at-risk individuals can deliver strong outcomes. After all, it’s far easier to catch a problem in its early stages than to deal with its serious downstream consequences. This is where devices that monitor patients continuously — outside the clinic, in real time — prove their worth most clearly.
A recent systematic review from JMIR, focused on wearables for chronic disease monitoring, finds that they are most widely used in cardiology, neurology, and metabolic disorders. The parameters being tracked include heart rate, body temperature, blood pressure, movement, respiration, and other physiological signals.
The American Diabetes Association’s 2025 Standards of Care lay out CGM as a key tool for continuous glucose monitoring, therapy adjustment, and lifestyle management. For millions of people, it provides a constant stream of data on how the body is behaving between clinic visits.
Much the same picture is emerging in cardiology. The Apple Heart Study, published in the NEJM, evaluated how Apple Watch’s irregular pulse notification feature could flag possible atrial fibrillation. The study enrolled more than 400,000 participants. Only 0.52% received an irregular pulse notification — rising to 3.2% among participants over 65. When those who were notified were then fitted with an ECG patch for follow-up verification, 34% went on to have atrial fibrillation confirmed. Moreover, when participants were wearing both the watch and the patch simultaneously, the rate of signal agreement came in at 84%. In other words, Apple Watch doesn’t hand down a diagnosis — but it can pick up on a suspicious rhythm with reasonable accuracy and flag that the person needs a proper clinical workup.
Leading Wearable Medical Device Companies and Startups
It makes sense to walk through some concrete device examples to get a real feel for what they’re actually capable of today. A clear trend stands out: the strongest players generally don’t try to measure everything at once. Instead, they zero in on a single scenario — monitoring blood sugar, arrhythmia, epileptic seizures, a patient’s condition after discharge, post-injury recovery — and refine their tracking to the point where the device can genuinely be used every day.
Abbott’s flagship wearable in this space is FreeStyle Libre. It’s a compact continuous glucose monitoring sensor that adheres to the skin and streams data to an app. Users can see exactly what’s happening to their glucose levels after meals, exercise, sleep, or stress.
The device has gone truly mainstream. Abbott states in its materials that FreeStyle Libre technology is used by millions of people worldwide, and in 2024 the company announced the expansion of its manufacturing facilities in Ireland to keep up with growing demand for FreeStyle Libre 3 sensors.
The best-known wearable medical device for the general public is, of course, Apple Watch — or more precisely, its pairing with the ECG app. The device doesn’t replace a full cardiological workup, and Apple is upfront about that. But the core idea proved powerful: a person can record an ECG resembling a single-lead reading straight from their wrist and check it for signs of atrial fibrillation. Apple notes that the ECG app is available on Series 4 and later (excluding SE) and can generate a recording similar to a single-lead electrocardiogram.
What makes Apple particularly interesting here goes beyond the ECG feature itself. The company has shown the market how a consumer device can slowly, step by step, close in on a genuine clinical use case.

BioIntelliSense’s BioButton is a clinical wearable. The device is a compact medical sensor that attaches to the chest and monitors a patient’s condition on a near-continuous basis. It tracks several key parameters simultaneously — including temperature, respiratory rate, heart rate, activity level, and body position — and streams that data to a digital platform where clinicians can review it. The device’s purpose isn’t to diagnose on its own, but to pick up on signs of deterioration as early as possible.
It’s a smart medical patch that keeps tabs on a patient far more frequently than standard manual check-ins allow. If readings start trending in a worrying direction, the system can alert a physician or care team before the patient even realizes their condition is getting worse. That makes devices like this especially valuable in hospital settings, following discharge, and in any situation where someone’s health needs to be watched continuously — not just checked in on now and then.
In 2024, the company announced FDA clearance for the BioButton Multi-Patient wearable device — a rechargeable, reusable solution designed to automate vital sign collection and scale hospital virtual care programs.
The Empatica Embrace2 is built around a single, focused task: epilepsy monitoring and the detection of possible convulsive seizures. On its official product page, Empatica describes Embrace2 as the first FDA-cleared wrist-worn wearable in epilepsy that detects possible convulsive seizures and immediately sends an alert to a caregiver. If a seizure occurs at home, at night, or when a person is alone, the device is designed to help summon help as quickly as possible.
Challenges Facing Wearable Medical Device Companies
Three core areas stand out where companies are focusing most of their development efforts.
The first challenge is signal quality and its interpretation. Any wearable sensor operates in “field conditions” — the person is walking, turning their wrist, sweating, sleeping in an awkward position, forgetting to charge the device, or simply wearing it differently than the manufacturer intended. Virtually everything affects the readings: how snugly the watch sits on the wrist, whether the sensor makes proper contact with the skin, whether arm movement introduces interference, or whether a weak signal or near-dead battery is skewing the data. So even when a device flags an anomaly, that doesn’t automatically make it a medical fact.

This is exactly why context matters just as much as the signal itself in wearable medicine. A one-off spike in heart rate after climbing stairs, downing a coffee, or going through a stressful moment is one thing. A sustained change that keeps recurring and is backed up by additional data is quite another. A device can send a genuinely useful alert — but that alert almost always needs to be followed up: with a repeat measurement, a cross-check against other readings, and sometimes a full clinical workup.
The second challenge is the often glaring gap between regulatory requirements and marketing promises. The FDA explicitly emphasizes on its digital health page that the list of cleared sensor-based digital health technology wearables is not exhaustive, and that the key determining factor remains the intended use — the stated purpose of the device.
In other words, a company’s advertising can hint at a great deal, while the regulator has cleared the device for a far narrower and more specific function. That’s where the disconnect comes from. Marketing sells the feeling of broad, sweeping benefit — almost a miniature medical revolution strapped to your wrist. But legally and clinically, the device may be approved for one specific task only, and with a long list of caveats. A watch may be able to flag possible signs of an irregular rhythm, but that’s not the same as it delivering a diagnosis. A patch may continuously gather vital signs, but that doesn’t mean it’s deciding what’s happening to the patient.
The third challenge is integration into clinical practice. Even a strong biosensor changes very little on its own if its data doesn’t feed into the real day-to-day workflow of a clinic. A device can continuously collect heart rate, temperature, respiration, activity levels, and other indicators — but that immediately raises the question of who, exactly, will be looking at all of this, how often, and what should happen when the system picks up a concerning deviation. If a physician already has an overloaded schedule and nurses don’t have dedicated time to work through a stream of notifications, even a genuinely useful device quickly turns into a source of extra noise.
That’s why it matters that the data flows into a clear digital system — one where it isn’t lost, duplicated, or siloed — and where it’s visible in the places where people act on it: an electronic health record, a clinical monitoring dashboard, or a remote patient monitoring platform.
There’s another dimension to this challenge as well: defining upfront what actually counts as an alert-worthy signal. If the system is too sensitive, it starts burying clinical staff in notifications, many of which will be false positives or clinically insignificant. If the thresholds are set too loosely, it risks missing a dangerous deterioration.
The success of wearables in medicine, then, depends not only on sensor accuracy, but on the organizational ecosystem built around it. It takes staff training, clear protocols, digital infrastructure, time set aside for data interpretation, and a real institutional readiness to absorb a new stream of information into everyday clinical work. And this is precisely the stage where many projects start to stall: building a good sensor is hard enough, but constructing a truly functional clinical workflow around it is often harder still.


